Overview
On February 19, 2026, the Court of Appeals for the Federal Circuit reinforced the principle that an ANDA label recommending an infringing use is not, by itself, sufficient to establish direct infringement of method claims.
The Patents and the Label
Corcept's patents claim methods of coadministering mifepristone (up to 900 mg) with strong CYP3A inhibitors to treat Cushing's syndrome. Teva's proposed generic label was essentially identical to Corcept's own 2019 Korlym label, which included dosing adjustments for administering up to 900 mg of mifepristone alongside strong CYP3A inhibitors — language that tracked the asserted method patents.
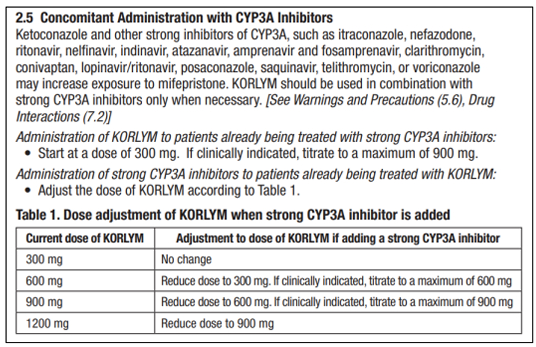
The Label Alone May Be Insufficient When Physician Practice Does Not Support Infringement
The District Court followed the Federal Circuit's earlier opinion in Genentech v. Sandoz decision, which held that a proposed label that "encourages, recommends or promotes an infringing use without any additional evidence showing such an infringing use will in fact occur, is insufficient for a finding of direct infringement." In other words, even a label that appears to instruct doctors to perform the patented method does not on its own establish that anyone will actually perform it.
The District Court looked beyond the label to evidence of actual physician behavior and clinical practice, asking whether doctors would actually use the patented methods if Teva's product came to market. The evidence showed they would not, for three reasons:
- Physicians consistently avoid coadministering mifepristone with strong CYP3A inhibitors due to documented safety concerns and dosing complexity.
- A non-infringing alternative (osilodrostat) was "available and preferred."
- A physician could follow Teva's label and still not infringe the claims, meaning the label did not compel infringement even if followed.
Equally notable, Corcept presented no evidence that any physician had ever practiced the claimed methods in the past, which the district court used as a permissible "starting point" for assessing future likelihood.
The Federal Circuit "perceived no error" in the District Court's analysis.
Practical Implications
The decision underscores a meaningful evidentiary burden, consistent with earlier Federal Circuit jurisprudence, on patent holders in Hatch-Waxman litigation. Pointing to label language that mirrors the claimed method is an important factor, but is not dispositive of the infringement analysis. The patentee must affirmatively demonstrate that the infringing use will actually occur in clinical practice. Where physicians have independent clinical reasons to avoid the patented method, or where non-infringing alternatives exist, courts may find that the label's instructions would not lead to actionable infringement.
Interestingly, the Court suggested that the entire dispute could have been avoided had Teva filed a "section viii carve-out" to remove the relevant use and dosing instructions from its label — a procedural tool that generics can use to sidestep patents covering specific methods of use while still bringing their product to market.
The content of this article is intended to provide a general guide to the subject matter. Specialist advice should be sought about your specific circumstances.




