- within Government, Public Sector, International Law and Real Estate and Construction topic(s)
- in United States
- with readers working within the Automotive, Consumer Industries and Oil & Gas industries
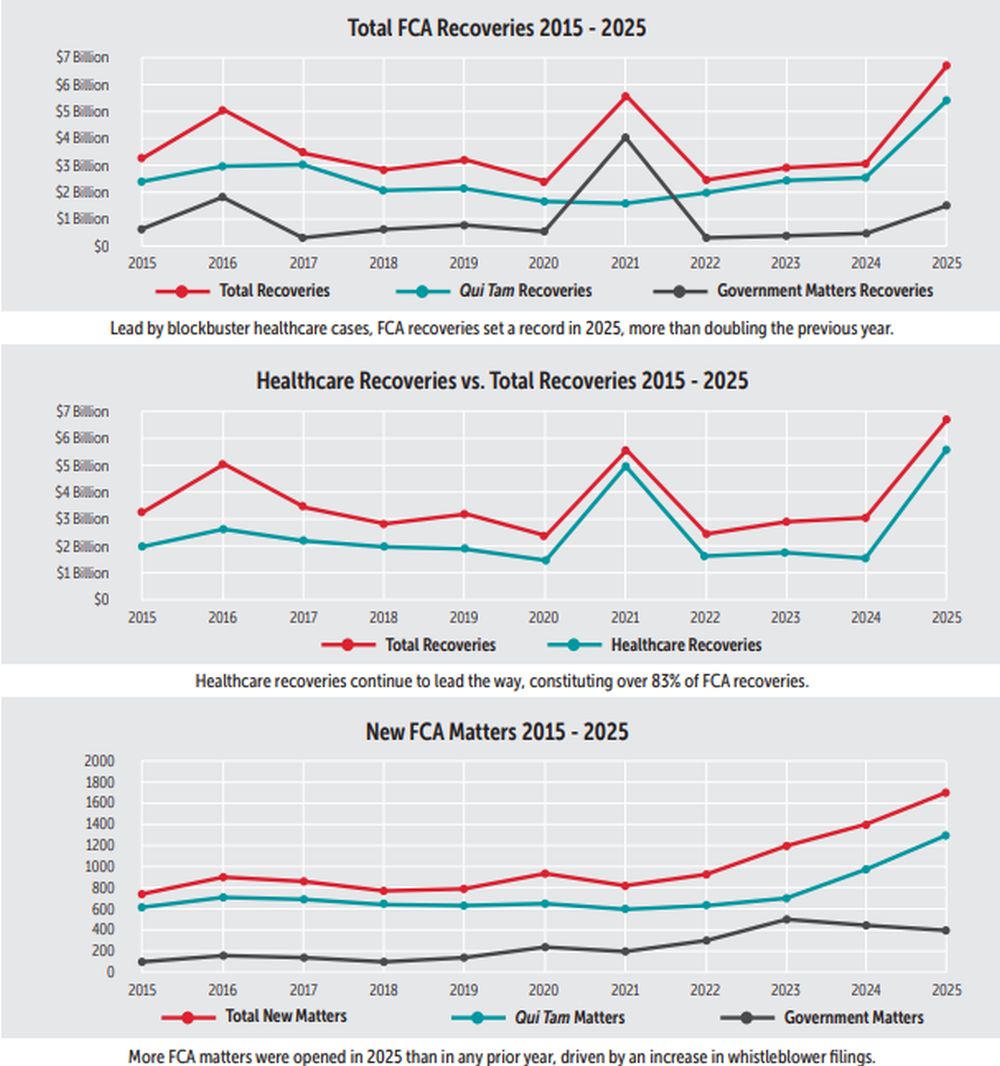
In fiscal year 2025, False Claims Act (FCA) recoveries reached their highest mark ever — over $6.8 billion. That total bests the prior record ($6.1 billion) set in fiscal year 2014, and more than doubles the single-year recoveries in any of the last three years. There were also more cases filed — and more cases filed by relators — than ever before. Interestingly, however, the total share of relators' awards went down, notwithstanding the rising cases and recoveries generally.
While 2025 was an outlier among the last several years, in some ways it mirrored the same trends we have come to expect. For example, the healthcare industry continued to be far and away the most targeted industry for FCA actions. Over $5.7 billion of the total — nearly 84% — arose from judgments and settlements involving healthcare industry defendants. Other areas of focus included tariff and custom duties, cybersecurity, and COVID-19-related fraud cases.
The year did not offer a watershed FCA opinion in the appellate courts. The sole Supreme Court opinion involving FCA issues was relatively narrow. It came in February in Wisconsin Bell, Inc. v. United States ex rel. Heath, 604 U.S. 140 (2025), in which the Court resolved a circuit split over what constituted a "claim" in the context of obtaining reimbursements from a telecommunication subsidy program.
Meanwhile, lower courts wrestled with many typical FCA issues. FCA liability premised on AntiKickback Statute (AKS) violations continued to be a favored theory by the government, and courts continued to address the appropriate causation standard in such cases. Scienter challenges, in the wake of the Supreme Court's 2023 decision in United States ex rel. Schutte v. SuperValu Inc., also continued to wind through the courts. Perhaps the biggest looming issue was heard at the end of the year, when the Eleventh Circuit held oral argument in United States ex rel. Zafirov v. Florida Medical Associates, LLC — the appeal from Judge Kathryn Mizzelle's decision in the Middle District of Florida finding that the FCA's qui tam provisions were unconstitutional.
But we will have to wait until next year's issue for the result of that case. For now, the pages that follow analyze the key cases and developments from 2025 and trends likely to come. As we enter the second year of the new administration, FCA enforcement remains robust, and FCA jurisprudence continues to be an ever-dynamic area of the law.
DOJ YEAR-END STATS
In fiscal year 2025, FCA recoveries topped $6.8 billion. The charts below and throughout the FCA Year in Review track notable trends in recoveries and other key metrics over the last decade.
KEY DECISIONS & DEVELOPMENTS
WHAT IS A CLAIM?
Wisconsin Bell, Inc. v. U.S. ex rel. Heath, 604 U.S. 140 (Feb. 21, 2025)
The Supreme Court resolves a circuit split, holding that requests for reimbursement under the FCC's "E-Rate" program qualify as claims under the FCA.
Relator Todd Heath alleged that common carrier Wisconsin Bell violated the "E-Rate" program, which subsidizes internet and other telecommunications services for schools and libraries across the United States. According to Heath, Wisconsin Bell did so by charging more for those services than allowed under the "lowest corresponding price" (LCP) rule set out in the Telecommunications Act of 1996 and its implementing regulations. The higher front-end price led to higher reimbursement requests to the FCC under the E-Rate program, and the reimbursements were paid out of the FCC's Universal Service Fund. Heath argued that this scheme caused reimbursement requests for amounts higher than the E-Rate program should have had to pay.
The trial court granted summary judgment to Wisconsin Bell on falsity and scienter grounds, holding that Heath failed to show (1) that Wisconsin Bell falsely certified compliance with the LCP rule; and (2) even if its certification was false, it was an honest misreading of the LCP rule rather than a knowing submission of false claims. The Seventh Circuit reversed, holding that Heath had presented sufficient evidence of falsity and scienter to proceed to trial. That court also considered two issues for the first time on appeal: (1) whether a jury could find that the alleged falsity of the claims was material to the government's payments; and (2) whether the allegedly fraudulent reimbursement requests for subsidies under the E-Rate program even amount to "claims" under the FCA. The court answered both questions in the affirmative.
The Supreme Court granted certiorari on the claims issue, citing a split between the Fifth and Seventh circuits. At the outset, the Court emphasized that "[t]he E-Rate reimbursement requests at issue count as FCA 'claims' if the Government 'provides or has provided any portion of the money requested.'" With that in mind, the Court echoed the Seventh Circuit's observation that the government has put substantial money into the Universal Service Fund. The Court therefore rejected Wisconsin Bell's argument that the fund consisted entirely of privately provided money. On that basis, the Court resolved a circuit split on the claims issue and determined that Heath's FCA suit could proceed to trial.
qwe
SCIENTER
To satisfy the FCA's scienter element, a defendant must either have actual knowledge of the falsity of information, act in deliberate ignorance of its truth or falsity, or act in reckless disregard of its truth or falsity.
U.S. ex rel. Sisselman v. Zocdoc, Inc., Case No. 24-2807 (2d Cir. Apr. 14, 2025)
The Second Circuit affirms lack of AKS or FCA scienter when corporation previously obtained OIG advisory opinions stating corporation's fee structure presented a low risk of fraud.
Zocdoc is an online tool that allows patients to search for and book appointments with various physicians. Physicians pay an annual fee to be listed on the website, and many also pay a "new-patient fee" anytime a new patient uses Zocdoc to book an appointment. If a physician does not pay new-patient fees, new patients cannot book appointments with them through Zocdoc, and the physician's placement or "ranking" in patient search results is affected. Relator Stephen Sisselman — a physician who used Zocdoc but only paid new-patient fees up to a pre-set monthly cap — alleged that Zocdoc's new-patient fees amounted to referral fees in violation of the AKS and FCA.
Zocdoc had previously obtained two advisory opinions from the Office of the Inspector General for the Department of Health and Human Services regarding its fee structure. The opinions stated that Zocdoc's new-patient fees "implicated" but did not violate the AKS, noting that higher fees did not result in better placement in patient search results and that fee amounts were not directly based on "the value of Federal health program business" Zocdoc generated. The U.S. District Court for the Southern District of New York dismissed Sisselman's claims, finding that Zocdoc's applications for advisory opinions demonstrated an effort to comply with applicable law and that Zocdoc did not mislead the OIG or act inconsistently with its opinions. Accordingly, Zocdoc did not possess the scienter required to violate the AKS or FCA. On appeal, the Second Circuit agreed, finding that Sisselman's complaint did not allege any facts to raise a "strong inference of fraudulent intent."
U.S. ex rel. Omni Healthcare Inc. v. MD Spine Solutions LLC, 160 F. 4th 248 (1st Cir. Dec. 1, 2025)
First Circuit holds as a matter of first impression that a laboratory can rely on a doctor's order to establish a rebuttable presumption that a test was "reasonable and necessary" at the summary judgment stage.
Relator Omni Healthcare sued MD Spine Solutions (MD Labs), a laboratory specializing in UTI testing, alleging that the laboratory was recouping payment for a more expensive test that was not "reasonable and necessary," thereby resulting in MD Labs knowingly submitting false claims to the government. The district court granted summary judgment in favor of MD Labs finding that it had provided sufficient evidence that the test was superior to the cheaper alternative, and that Omni, the medical practice that ordered the tests, had failed to provide any evidence to the contrary to submit as a triable issue of fact. Omni appealed.
The First Circuit agreed with the district court and held as a matter of first impression that in FCA cases alleging Medicare fraud against a laboratory, the laboratory can rely on a doctor's order to show that a test was "reasonable and necessary." The burden then shifts to the FCA claimant to provide evidence of scienter to rebut this presumption. The First Circuit reasoned that while doctors' offices work directly with patients, a laboratory completing the requisite tests lacks both the expertise and discretion to diagnose patients, thus making a doctor's order a reliable safe harbor to establish medical necessity. Accordingly, the First Circuit affirmed the district court's grant of summary judgment in favor of MD Labs. For further analysis of the Omni case, please see this recent blog post1 on Bradley's Eye on Enforcement blog.
To view the full article, click here.
Footnote
The content of this article is intended to provide a general guide to the subject matter. Specialist advice should be sought about your specific circumstances.


