- within Antitrust/Competition Law topic(s)
Amendment in Handbook of Procedure 2023 with regard to Letter of Permission for Export Oriented Units (EOUs), Electronics Hardware Technology Parks (EHTPs), Software Technology Parks (STPs) and BioTechnology Parks (BTPs)
The Directorate General of Foreign Trade has amended the validity period of Letter of Permission (LOP) / Letter of Intent (LOI) for Export Oriented Units, Electronics Hardware Technology Parks, Software Technology Parks, and BioTechnology Parks, to streamline the administrative processes. The revision is as follows:
- Validity period of LOP / LOI once unit commences production be extended for a period of up to five years at a time or;
- The two years initial validity of LOP / LOI (except in case where there is a restriction on the initial period of approval, like setting up of oil refinery project) will be extended by one additional year for valid reasons to be recorded in writing.
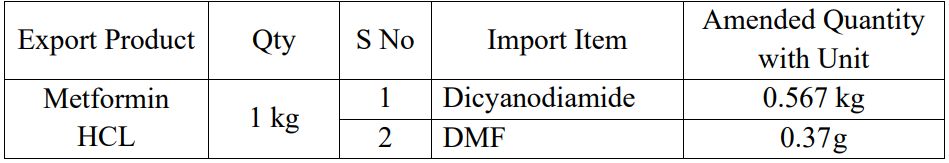
Amendments in Standard Input Output Norms for Metformin (11 Dec)
The Directorate General of Foreign Trade has amended the Unit of Measurement used in Standard Input Output Norms for Metformin HCL.
Revised SIONs for the Chemical and Allied Products (17 Dec)
The Directorate General of Foreign Trade has amended the Standard Input Output Norms (SIONs) for Chemical & Allied Products and General Note for all export product groups stands amended with immediate effect. The list of revised Standard Input Output Norms is provided in the link herein.
Amendment in Import Policy Condition for items covered under Chapter 29 (18 Dec)
The Directorate General of Foreign Trade has amended the Import Policy Condition for Chapter 29 with immediate effect, to prescribe minimum CIF value, applicable till 30th November 2026. The revisions are as follows.
- Imports of diluted Potassium Clavulanate having a CIF value of less than USD 77 per Kg shall be 'Restricted'.
- Imports of Potassium Clavulanate having a CIF value of less than USD 180 per KGA shall be 'Restricted'.
- Imports of the following intermediates used for the manufacture
of Clavulanic Acid or Potassium Clavulanate, having a CIF value
below USD 92 per KG shall be 'Restricted':
- Tertiary Butylamine Salt - Potassium Clavulanate;
- Tertiary Octylamine Salt - Potassium Clavulanate;
- Tertiary Butylamine Salt - Intermediate of Clavulanic Acid;
- Tertiary Octylamine Salt - Intermediate of Clavulanic Acid.
The complete notification is available at the link herein.
The content of this article is intended to provide a general guide to the subject matter. Specialist advice should be sought about your specific circumstances.
[View Source]

